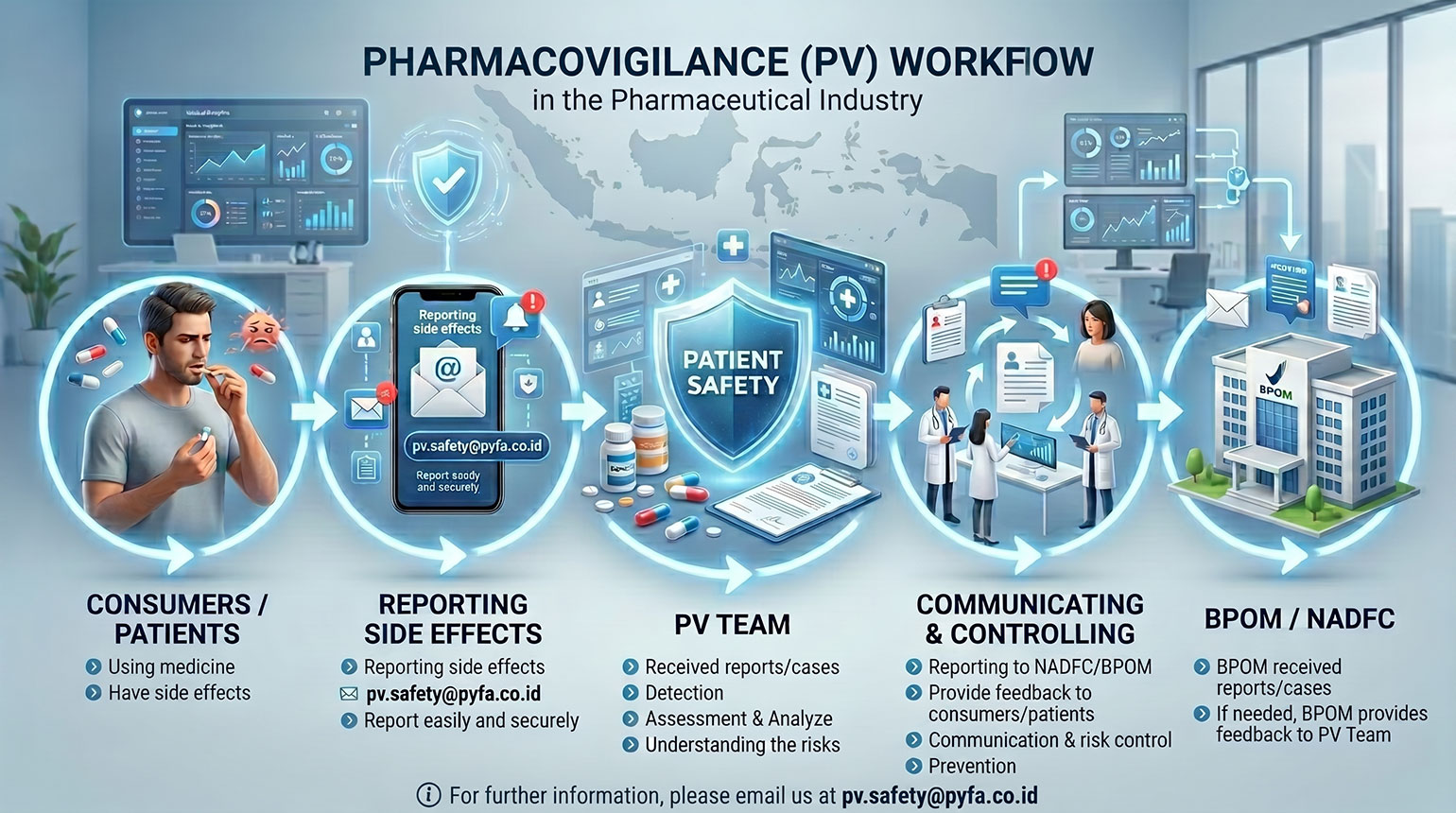
Pharmacovigilance
As a Marketing Authorization Holder (MAH), PT Pyridam Farma Tbk conducts Pharmacovigilance activities to ensure the safety of marketed products in compliance with applicable regulations, supported by a dedicated Pharmacovigilance department.
Pharmacovigilance is all activities of detecting, assessing, understanding, communicating, controlling and preventing side effects or other problems related to the use of drugs, natural medicines, health supplements, cosmetics and quasi-drugs